Test for COVID-19 to be done at home
Ten post jest także dostępny w języku:
![]() polski
polski
The California-based company Lucira Health received the first FDA approval to market the COVID-19 test to be performed by a patient at home. The result is to be known after only 30 minutes. This is to speed up the diagnosis of COVID-19 patients significantly.
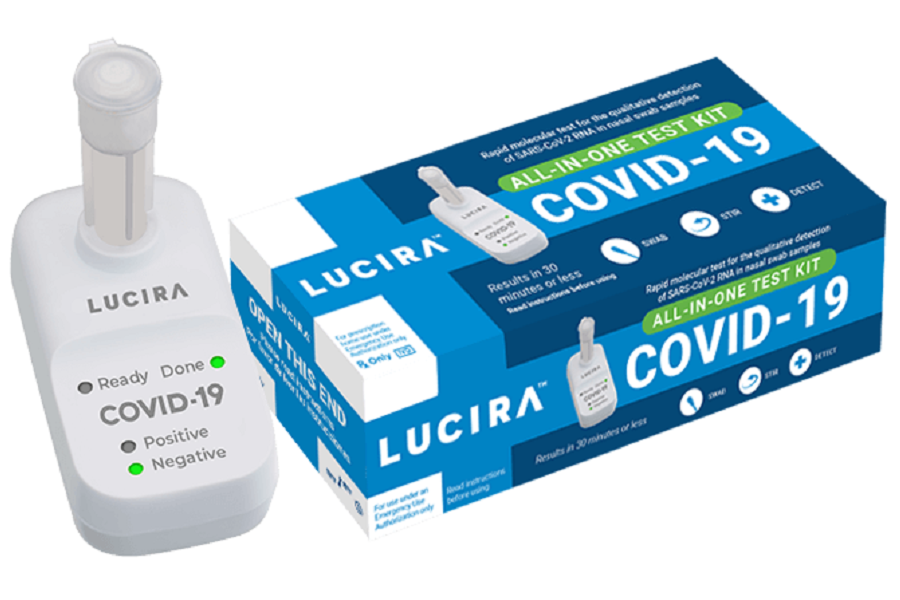
Approval of Lucira COVID-19 All-In-One Test Kit by FDA
Lucira Health has developed a test kit designed to detect SARS-CoV-2 coronavirus at home. After taking a nasal swab, the patient must place it in the reagent vial. Then the material prepared in this way is placed in the attached portable device and the information whether the patient is infected with COVID-19 is already known after 30 minutes. The company emphasizes that the test kit developed for COVID-19 is an in vitro molecular diagnostic test that has an analytical sensitivity or SARS-CoV-2 detection capability comparable to some molecular tests used by laboratories.
The Lucira COVID-19 All-In-One test kit is the first FDA-approved prescription test in an emergency procedure (EUA) to be performed at home for people aged 14 and over. The company conducted a study in the summer of 2020 on a sample of 352 people, which showed that 100% of the test users aged 14 years and over were able to perform the test correctly at home.
Photo: www.lucirahealth.com