Novartis will introduce the world’s most expensive medicine to the market
Ten post jest także dostępny w języku:
![]() polski
polski
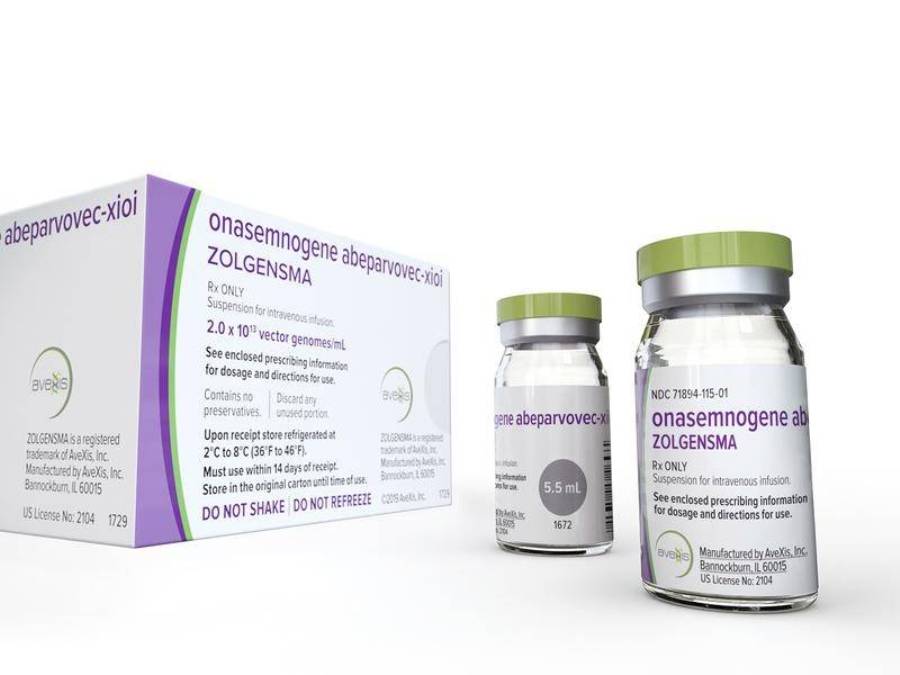
AveXis, a company belonging to the Swiss concern Novartis, announced on Friday that it had received the FDA’s approval for the marketing of ZolgenSMA. It is a genetic therapy used in children under 2 years of age suffering from spinal muscular atrophy (SMA).
The cost of ZolgenSMA is $2.125m
ZolgenSMA therapy (onasemnogene abeparvovec) consists in replacing the damaged SMN1 gene with its efficient copy. This is done by a single infusion, which is able to stop the development of the disease. The cost of such an infusion is over $2m. However, as Novartis points out, the cost of current therapy for the disease in the first 10 years of a child’s life is over $4m. And such treatment must be carried out throughout life.
ZolgenSMA is currently registered in the USA, but AveXis also intends to register the drug in European countries and Japan. The company is also working on a preparation administered through a lumbar puncture, which could be used in older children and adults.
Will ZolgenSMA be reimbursed in Poland?
SMA, or spinal muscular atrophy, is a neuromuscular disease with a genetic basis. The disease is particularly rapid in infants and young children and is the most common genetic cause of death in this age group. In total, according to SMA Foundation data, there are about 400-800 people suffering from the disease in Poland.
Worthy of note, since January 2019 Spinraza (nusinersen) has been included in the reimbursement system in Poland. The drug produced by Biogen slows down the development of SMA. It is administered as part of the drug programme. The catalogue price of one injection is €90,000, but the price negotiated by the Polish payer may be several times lower. In the first year of therapy, the patient must receive six injections, and in each subsequent year – three. Will the Polish patients also be reimbursed with ZolgenSMA?