PZ Cormay to distribute coronavirus tests in Poland
Ten post jest także dostępny w języku:
![]() polski
polski
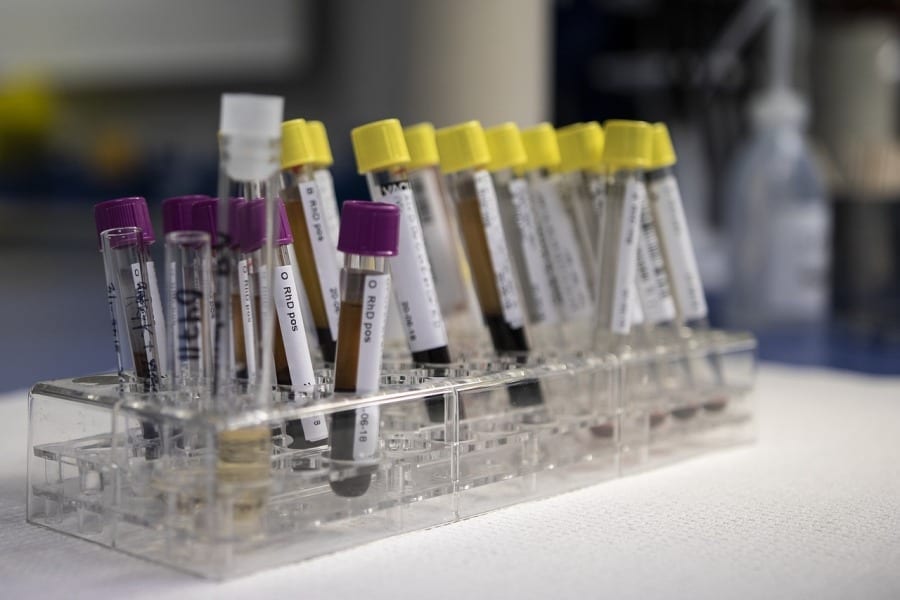
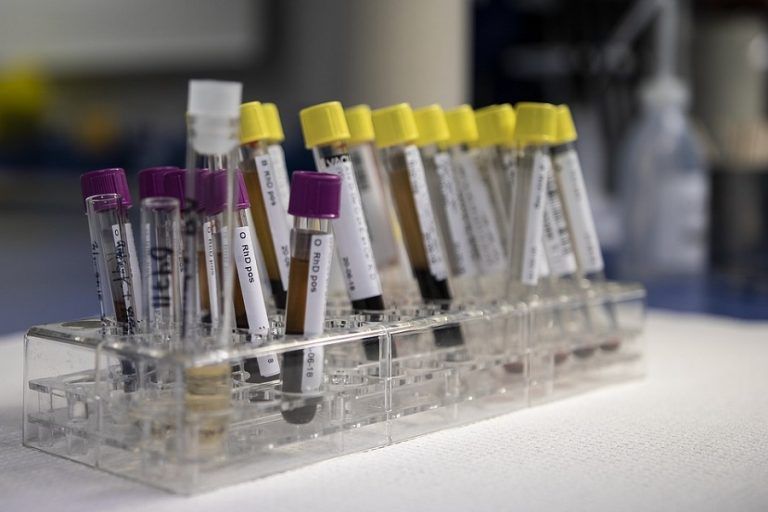
PZ Cormay, a Polish manufacturer of reagents for laboratory diagnostics, will notify the Office for Registration of Medicinal Products, Medical Devices and Biocidal Products (URLP) of two immunological reagents detecting the presence of the coronavirus causing COVID-19 disease.
SNIBE’s tests available in the EU
These are Maglumi reagents, i.e. 2019-nCoV IgG and 2019-nCoV IgM. They are designed for immune diagnostics and enable marking the amount of IgG and IgM antibodies to the coronavirus. The tests were developed by Shenzhen New Industries Biomedical Engineering (SNIBE) and are already used in other countries. On 19 February SNIBE submitted the CE declaration, which opened the way for the use of the tests in the European Union. Among the first test buyers in the EU was Italy, where the number of infections is currently the highest in Europe.
Cormay: Several centres in Poland can diagnose COVID-19
According to Cormay, the tests are designed to be used under laboratory conditions and can be used in conjunction with other procedures (including current genetic tests available) to increase the effectiveness of diagnosis. It is worth noting that Maglumi tests are currently the only serological tests available on the market for COVID-19 diagnosis.
Submitting tests to the URPL enables the immediate start of sales in Poland. Currently only a few diagnostic centers in Poland have the equipment to perform tests with Maglumi reagents. Cormay is in talks with other diagnostic centers to equip them with appropriate analysers.





I was diagnosed of genital Herpes for 10 years, and ever since then i have been taking treatment to prevent outbreaks, burning and blisters, but there was no improvement until i came across testimonies of Dr. Timothy on how he has been curing different people from different diseases all over the world, then i contacted him. After our conversation he sent me the medicine which i took according to his instructions for up to 20 days. After completing the medication i went back to my doctor for another test and the virus was all gone and i was completely cured, since then i have not had any signs of outbreak. I’m so filled with joy. With herbal medication Herpes Virus is 100% curable. I refer Dr. Timothy to everyone out there with the virus. Email address is goodhealthherbalfoundation@yahoo.com OR web https://5dc41d5b6d60d.site123.me