PMR report: The number of companies on the biological drugs market is growing

Ten post jest także dostępny w języku:
![]() polski
polski
According to forecasts presented in the PMR report “Biological and biosimilar drugs market in Poland 2019. Market analysis and development forecasts for 2019-2024”, the market of reference biological drugs in Poland is growing much slower than that of biosimilar products. In 2018 it reached a growth rate of 3%, compared to 40% in the market of biosimilar drugs. This is mainly due to the high base and new launches of biosimilars, which are associated with the loss of patent protection by several leading original medicines.
Number of companies generating sales of biological drugs on the Polish market
In total, according to PMR data, the number of companies present on the biological drugs market in Poland in 2017-2018 remained at a similar level. This is due to the fact that barriers to market entry, due to the cost and length of development of a new drug, are very high. Only the first half of 2019 brought a slight increase in the number of players present on the market, which was largely due to allowing for the reimbursement of further biosimilar drugs, but also to entry into the pharmacy segment (e.g. in the case of Aflofarm).
It is worth noting that in terms of value, original biological products predominate in the Polish biological drugs market, mainly due to the much higher costs of such treatments. The large number of available products is also important. From 2017 to the first half of 2019, only 42 biosimilar products generated any sales in the Polish market. By comparison, there were six times as many reference drugs in the same period (for many of them there are no biosimilars).

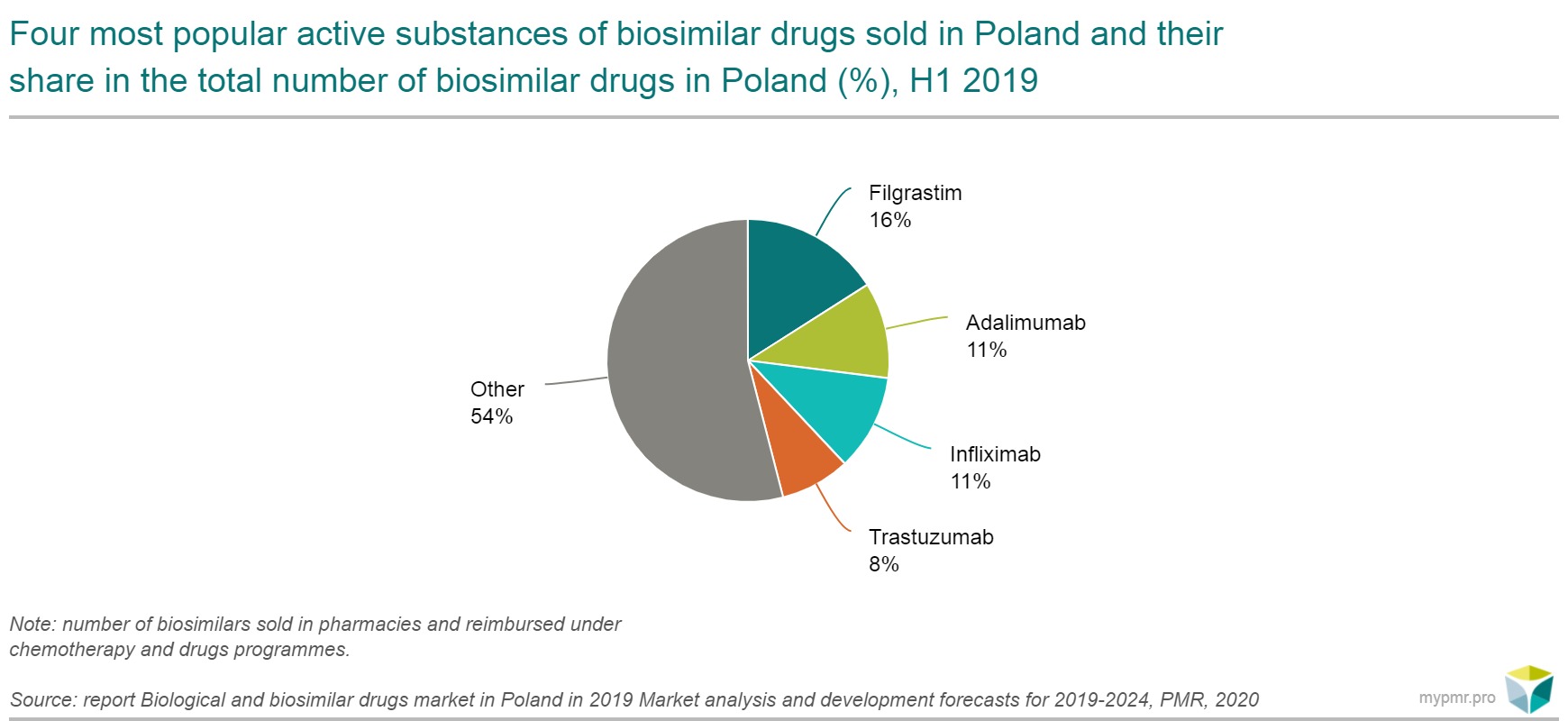
The most popular active substances of biosimilar drugs in Poland
According to PMR’s calculations, in Poland, in the first half of 2019, the most sales-generating biosimilar products (trade names) contained filgrastim, adalimumab and infliximab (substances with a share of over 10% in terms of the number of products). For adalimubab, the reference product Humira still accounted for the majority of sales. This was due to the policy of Abbvie, which reduced the price of its medicine to the price of its counterparts following the entry of biosimilar products for reimbursement in the first half of 2019.
According to data presented by the Modern Healthcare Institute, the reference price of adalimubab 40 mg (valid until the end of December 2018) was 53% higher than that of its biosimilar counterparts that appeared in the reimbursement in the first half of 2019. For Neupogen (reference filgrastim) and Remicade (reference infliximab), their share of the sales value of the active substance is currently marginal and fluctuates around 1 %.




I lost my Job few months ago,my wife left me ,could not get income for my family, things was so tough and I couldn’t get anything for my children, not until a met a recommendation on a page writing how Mr Bernie Wilfred helped a lady in getting a huge amount of profit every 6 working days on trading with his management on the cryptocurrency Market, to be honest I never believe it but I took the risk to take a loan of $1000. and I contacted him unbelievable and I was so happy I earn $12,500 in 6 working days, the most joy is that I can now take care of my family I don’t know how to appreciate your good work Mr. Bernie Doran God will continue to bless you for being a life saver I have no way to appreciate you than to tell people about your good services. For a perfect investment and good strategies contact Mr Bernie Doran via WhatsApp :+1(424)285-0682 or Telegram : @Bernie_fx or Gmail : Bernie.doranfx01@gmail.com